
Position
Professor, Department of Medicine, McGill University
Associate Member, Department of Microbiology & Immunology, McGill University
Strauss Chair in Respiratory Diseases
Associate Director, McGill International TB Centre
Research Interests
Our lab focuses on investigating the cellular and molecular mechanisms involved in the cross-talk between innate and adaptive immunity to major pulmonary infectious diseases, such as influenza and tuberculosis. Host survival during infection in vital organs, such as the lung, requires a delicate balance between host resistance, which is essential for detecting and eliminating the pathogens, and disease tolerance, which is critical in minimizing collateral tissue damage. We are also investigating how to harness the power of innate immunity in vaccine via reprogramming of hematopoietic stem cells. In summary, understanding the regulatory mechanisms involved in both host-resistance and tolerance is essential for the future development of an effective therapy or vaccine.
Research Foci
- Host defense (Resistance & Tolerance) against influenza and TB
- Hematopoietic Stem Cells in Infection and Vaccine
- Trained Innate Immunity
- Macrophage Cell Death Programs
- Metabolism in Infectious Diseases
- Eicosanoids in Infectious Diseases
Article in International Innovation
*International Innovation, published by Research Media, is the leading global dissemination resource for the wider scientific, technology and research communities, dedicated to disseminating the latest science, research and technological innovations on a global level. More information and a complimentary subscription offer to the publication can be found at: www.researchmedia.eu
Podcast with Dr. Divangahi
Causes or Cures is a health podcast hosted by Dr. Eeks.
Listen to the July 15, 2025 podcast with Dr. Divangahi:
Can Mushroom Fiber Beat the Flu? Beta-Glucan 101 with Dr. Maziar Divangahi. Episode 234
Featured Publications
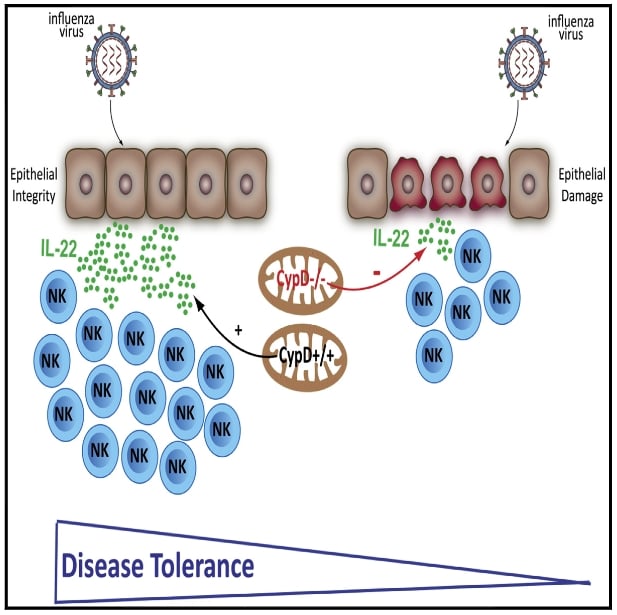
Mitochondrial cyclophilin D promotes disease tolerance by licensing NK cell development and IL-22 production against influenza virus
Downey J, Randolph HE, Pernet E, Tran KA, Khader SA, King IL, Barreiro LB, Divangahi M. Cell Rep. 2022 Jun 21;39(12):110974.

Brain motor and fear circuits regulate leukocytes during acute stress
Poller WC, Downey J, Mooslechner AA, Khan N, Li L, Chan CT, McAlpine CS, Xu C, Kahles F, He S, Janssen H, Mindur JE, Singh S, Kiss MG, Alonso-Herranz L, Iwamoto Y, Kohler RH, Wong LP, Chetal K, Russo SJ, Sadreyev RI, Weissleder R, Nahrendorf M, Frenette PS, Divangahi M, Swirski FK. Nature. 2022 Jul;607(7919):578-584.

Lack of evidence for intergenerational inheritance of immune resistance to infections
Kaufmann E, Landekic M, Downey J, Chronopoulos J, Teimouri Nezhad S, Tran K, Vinh DC, Barreiro LB, Divangahi M. Nat Immunol. 2022 Feb;23(2):203-207.

Training the metaorganism: the microbial counterpart
King IL, Divangahi M. Cell. 2021 Jan;184(3):574-576.

Trained immunity, tolerance, priming and differentiation: distinct immunological processes
Divangahi M, et al. Nat Immunol. 2021 Jan;22(1):2-6.

M. tuberculosis Reprograms Hematopoietic Stem Cells to Limit Myelopoiesis and Impair Trained Immunity
Khan N, Downey J, Sanz J, Kaufmann E, Blankenhaus B, Pacis A, Pernet E, Ahmed E, Cardoso S, Nijnik A, Mazer B, Sassetti C, Behr MA, Soares MP, Barreiro LB, Divangahi M. Cell. 2020 Oct 29;183(3):752-770.e22.

Editorial: Evolving Mechanisms of Disease Tolerance
King IL, Divangahi M. Front Immunol. 2019 Dec 20;10:2974.

Leukotriene B4-type I interferon axis regulates macrophage-mediated disease tolerance to influenza infection
Pernet E, Downey J, Vinh DC, Powell WS, Divangahi M. Nat Microbiol. 2019 Aug;4(8):1389-1400.
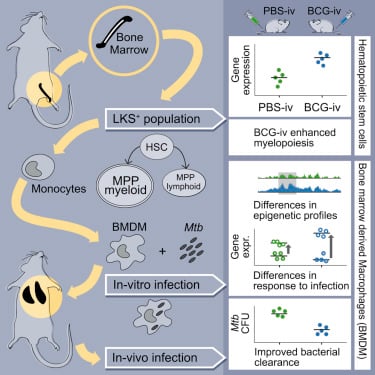
BCG Educates Hematopoietic Stem Cells to Generate Protective Innate Immunity against Tuberculosis
Kaufmann E, Sanz J, Dunn JL, Khan N, Mendonça LE, Pacis A, Tzelepis F, Pernet E, Dumaine A, Grenier JC, Mailhot-Léonard F, Ahmed E, Belle J, Besla R, Mazer B, King IL, Nijnik A, Robbins CS, Barreiro LB, Divangahi M. Cell. 2018 Jan 11;172(1-2):176-190.e19.
In the News

Canada Research Chair in Trained Immunity
Maziar Divangahi appointed a new Canada Research Chair in Trained Immunity

2026 RESP Research Day Poster and Oral Winners
Congratulations to all of our oral and poster presenter winners from the 2026 Respiratory Research Day

CIHR Team Grant: Bringing Biology to Cancer Prevention
Congratulations to Dr. Maziar Divangahi and his team on receiving a $2M CIHR team grant to study trained immunity and cancer risk.

CIHR Project Grant Results – Fall 2025
Congratulations to all RESP members who were funded in the Fall 2025 CIHR Project Grant Competition!

β-glucan and Dietary Supplements
Maziar Divangahi discusses his research on β-glucan and lung inflammation with Radio-Canada

Philanthropy Fuels Innovation: 2025 MGH Research Awards
Several RESP Program researchers are among the recipients of the Montreal General Hospital Foundation’s 2025 Research Awards

CIHR Project Grant Results – Spring 2025
Congratulations to all RESP members who were funded in the Spring 2025 CIHR Project Grant Competition!

Trained Immunity May Cause Lung Damage
New study by Maziar Divangahi reveals that trained immunity may worsen lung inflammation during severe inflammation caused by viral or bacterial products

Breakthrough to Boost Bladder Cancer Treatment
Research led by Maziar Divangahi reveals how B-glucan can boost the effectiveness of BCG therapy for bladder cancer

Sustaining Excellence: Supporting Breakthrough Research
Larry Lands, Arnold Kristof, Salman Qureshi, and Maziar Divagnahi receive funding from the Sustaining Excellence Grants Competition.

Spring 2025 Student Awards
Congratulations to the 2025 recipients of FRQS and CIHR studentship and fellowship awards! We wish you much success!

2025 RESP Research Day Poster and Oral Winners
Congratulations to all of our oral and poster presenter winners from the 2025 Respiratory Research Day

Beta-glucan can ‘Reprogram’ Immune Cells With Promising Results
A study by Maziar Divangahi found that beta-glucan, a component in all fungi, can protect against flu-related lung damage.

Pregnancy’s Secret Flu Shield
Maziar Divangahi and James Martin uncover a natural flu defense that activates in the nasal cavity during pregnancy

2024 MGH Research Awards
Several RESP Program researchers are among the recipients of the Montreal General Hospital Foundation’s 2024 Research Awards

CIHR Project Grant Results – Spring 2024
Congratulations to all RESP members who were funded in the Spring 2024 CIHR Project Grant Competition!

New 2024 Grants to Meakins Faculty
Congratulations to Irah King, Dao Nguyen, Maziar Divangahi, and Gregory Fonseca on their recent grants!

2024 RESP Research Day Poster and Oral Winners
Congratulations to all of our oral and poster presenter winners from the 2024 Respiratory Research Day.
Contact Information
Meakins-Christie Laboratories
RI-MUHC, Block E
Office EM2.2248
Lab E03.2379
1001 Decarie Blvd.
Montreal QC H4A 3J1
Canada
Tel: 514-934-1934
E-Mail: maziar.divangahi [at] mcgill.ca
Education & Training
BSc (Biol) McMaster, 2000
PhD (Immunol), McGill, 2005
PDF (Inf Dis), McMaster, 2007
PDF (Innate Immunity), McGill, 2008
PDF (Macrophage Death), Harvard, 2010
Teaching
Biochemical Pathology (Cytokine biology), PATH 607